
In this respect, besides the canonical cct and cpn60 genes described above, three divergent hsp60-like genes have been more recently identified in association with pathological conditions. Īs for other chaperones, the malfunctioning of chaperonin proteins has been associated with various human pathological conditions, the chaperonopathies. The CCT complex is mostly known for its role in folding the cytoskeleton proteins actin and tubulin and mutations in individual CCT subunits lead to defects in the functioning of the cytoskeleton and mitosis arrest. Eukaryotic Group II chaperonin proteins assemble in a similar double-ringed oligomeric structure, called TRiC or CCT complex, composed of 16 subunits that in human are encoded by nine distinct genes ( tcp1/ cct1, cct2-5, cct6A-B, cct7-8). The Group I chaperonins are typically formed by the products of a single gene ( groEL in bacteria hsp60/ cpn60 in mitochondria) assembled into a 14-subunit double-ringed structure in bacteria and into a double or single-ringed structure in mitochondria. Chaperonin proteins form typical multi-subunit double-ringed structures collectively called "chaperonins". In eukaryotes, Group I chaperonins are mostly expressed in mitochondria and chloroplasts, and Group II chaperonins are found in the eukaryotic cytosol. The eukaryotic cell expresses representatives of two distinct groups of chaperonin genes that are otherwise typical of bacteria (Group I) or archaea (Group II). Hsp60-like chaperonin proteins are well known for their role in assisting protein folding and in protecting cells from the deleterious effects of stress. The functional characterization of newly identified chaperonin genes will be a challenge for future experimental analyses. In vertebrates, chaperonin genes, driven by intense duplication processes, have diversified into multiple classes and functionalities that extend beyond their well-known protein-folding role as part of the typical oligomeric chaperonin complex, emphasizing previous observations on the involvement of individual CCT monomers in microtubule elongation. The characterization of many newly-discovered chaperonin pseudogenes uncovered the intense duplication activity of eukaryotic chaperonin genes. Detailed sequence analysis and structural predictions of MKKS, BBS and CCT8L proteins strongly suggested that they conserve a typical chaperonin-like core structure but that they are unlikely to form a CCT-like oligomeric complex. 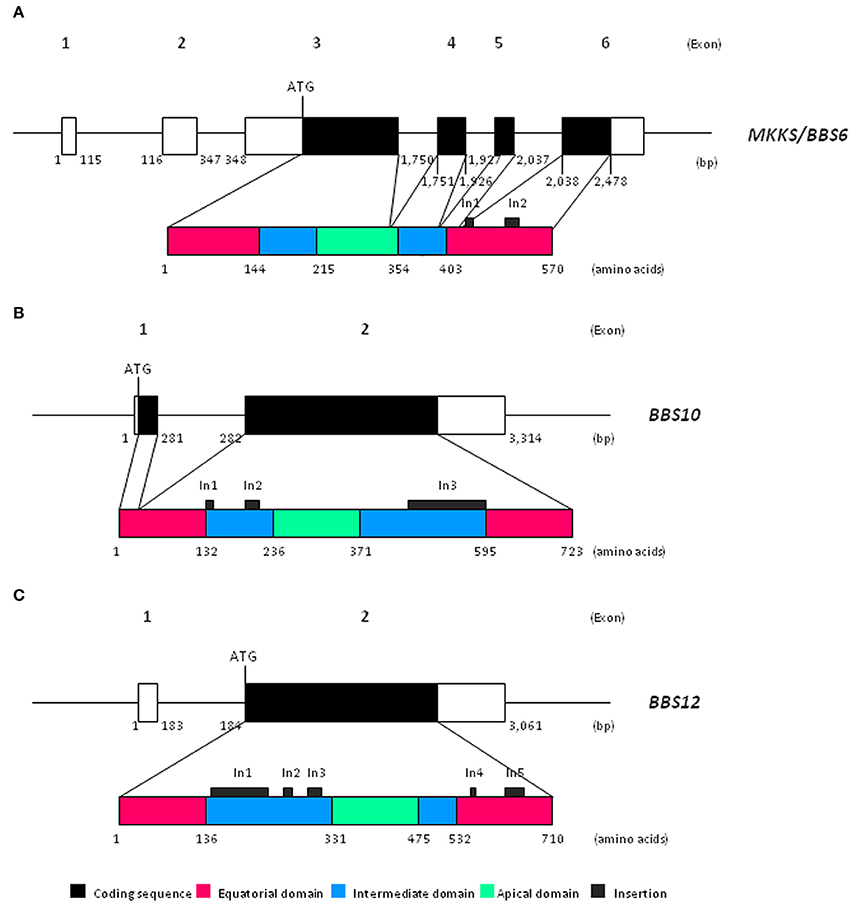
The functionality of CCT8L genes in different species was confirmed by evolutionary analyses and in human by expression data. The CCT8L gene originated from a later duplication also in the CCT8 lineage at the onset of mammal evolution and duplicated in primate genomes. Comparative analyses from several vertebrate genomes established the monophyletic origin of chaperonin-like MKKS and BBS genes from the CCT8 lineage. 
In mammal genomes we identified, besides the well-known CCT chaperonin genes and the three genes associated with the MKKS and BBS pathological conditions, a newly-defined class of chaperonin genes named CCT8L, represented in human by the two sequences CCT8L1 and CCT8L2. We identified fifty-four chaperonin-like sequences in the human genome and similar numbers in the genomes of the model organisms mouse and rat. The availability of complete genome sequences makes possible a definitive characterization of the complete set of chaperonin sequences in human and other species. However, the recent identification of three diverged chaperonin paralogs associated with the human Bardet-Biedl and McKusick-Kaufman Syndromes (BBS and MKKS, respectively) indicates that the eukaryotic chaperonin-gene family is larger and more differentiated than previously thought. Chaperonin proteins are well known for the critical role they play in protein folding and in disease.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
